Oncology trials face unique recruitment challenges despite millions of cancer patients worldwide. Explore the five biggest barriers in oncology clinical trials and strategies to improve enrollment.
TL; DR (for the busy sponsors & Onco experts)
- Oncology trials don’t lack patients; they lack access to the right patients at the right time. Strict eligibility criteria, biomarker requirements, and rapid disease progression significantly shrink the recruitable population.
- Many eligible patients never hear about clinical trials. Physician time constraints, limited awareness of available studies, and institutional barriers can prevent trial discussions early in the patient’s journey.
- The critical recruitment window occurs before treatment begins. The 4–12 weeks (variable) between diagnosis and therapy decisions is often the only opportunity for patients to consider clinical trials.
- Early patient navigation can transform recruitment outcomes. Providing guidance, education, and coordination during this window helps patients explore trial options while improving recruitment efficiency for sites and sponsors.
- Clariness’ OncoCONNECT solution helps reach patients earlier. By supporting patients between diagnosis and treatment decisions, trials can be introduced before recruitment opportunities are missed.

It is projected that there will be 28 million new cancer cases each year by 2040. At the same time, according to the Global Data, there are more than 32,000 ongoing and planned cancer clinical trials in 2026.
Despite the large number of patients and research efforts, oncology trials struggle to recruit participants. The challenge is not the absence of patients, but the difficulty of identifying and enrolling the right patients quickly enough. Cancer research moves rapidly, and many patients cannot afford to wait. Every delay in recruitment can mean missed opportunities for patients to access potentially life-changing therapies, while also slowing the progress of research.
Here are the five biggest recruitment challenges in oncology trials:
1. Extremely narrow eligibility criteria
Oncology trials frequently require highly specific patient profiles. In the US, only 3-5% of adult cancer patients (fewer than 1 in 20 patients worldwide) participate in clinical trials, mainly due to stringent eligibility criteria.
Eligibility may depend on factors such as the exact cancer subtype, disease stage, prior lines of therapy, and the presence of particular genetic mutations or biomarkers.
While these criteria are often necessary for scientific rigor and precision medicine approaches, they drastically reduce the pool of eligible participants. As a result, many patients who initially appear suitable are later excluded during screening. According to the Cancer Network, strict trial eligibility criteria can exclude up to 25% of patients who attempt to enroll in a study.
2. Physician gatekeeping
Oncologists play a central role in determining whether patients learn about clinical trial opportunities. In many cases, physicians may prioritize established standard-of-care treatments, particularly in first-line settings where proven therapies are available.
There are several reasons for this gatekeeping effect. Physicians may have limited time during consultations to discuss trial options, may not be aware of all active studies for a specific indication, or may worry about the potential impact of experimental treatments on their patients’ well-being.
As a result, many eligible patients are never informed about relevant clinical trials. Recruitment therefore becomes heavily dependent on physician engagement and awareness.
Additionally, referring patients to external trial sites may not always align with the financial or operational incentives of healthcare providers, further limiting patient referrals.

3. Biomarker & genetic testing barriers
Precision oncology increasingly relies on molecular profiling to identify patients who may benefit from targeted therapies. Many trials therefore require biomarker confirmation through genetic testing or fresh tumor biopsies.
However, these requirements introduce additional logistical challenges. Tumor biopsies can be invasive, and molecular testing may involve complex laboratory processes with turnaround times that can take several weeks.
These delays can significantly impact recruitment. Patients may become ineligible while waiting for test results, begin other treatments, or simply drop out of the process. In addition, the costs associated with advanced molecular testing can add further financial pressure for sponsors.
4. Rapid disease progression & limited time windows
Cancer can progress quickly, leaving a very limited window in which patients are eligible for clinical trial participation.
Between diagnosis, referral, screening, and eligibility confirmation, patients’ health status may deteriorate. In other cases, physicians may decide that immediate standard-of-care treatment is necessary, leaving little time to consider clinical trial enrollment.
This urgency creates a challenging recruitment environment. Even when patients initially qualify, they may become ineligible before enrollment can be completed, contributing to higher screen failure and dropout rates.
5. Competition between trials
Oncology is one of the most active areas in clinical research. For common indications such as breast, lung, or colorectal cancer, multiple studies may recruit simultaneously for very similar patient populations.
This creates intense competition for a limited pool of eligible participants. In many cases, several trials may be recruiting at the same hospitals or cancer centers, forcing sites to divide attention and resources between multiple sponsors.
For sponsors, this competition can lead to slower enrollment, longer timelines, and increased recruitment costs.

The gap in the patient journey
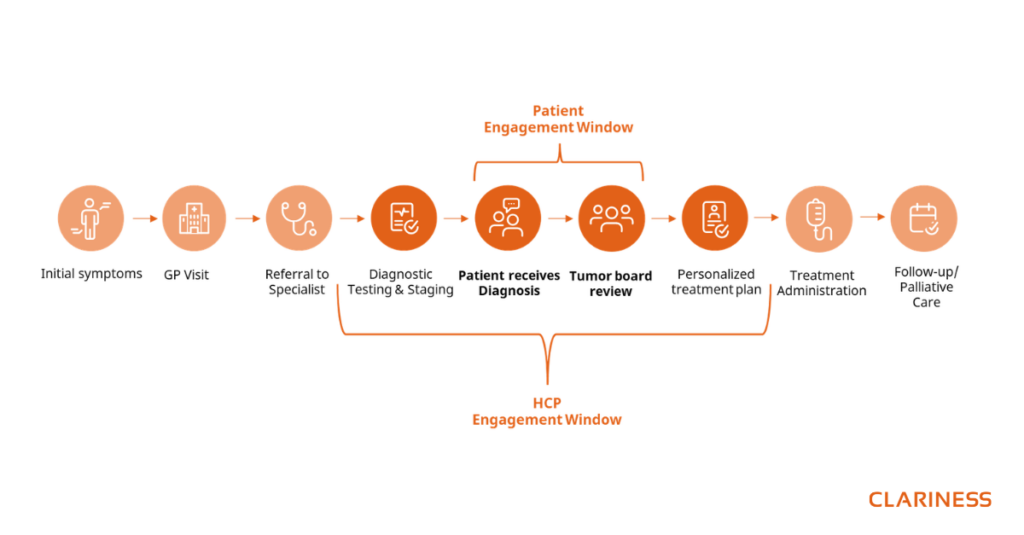
Many of the barriers in oncology trials occur before a patient reaches a clinical trial site. In fact, the short window between receiving a cancer diagnosis and starting treatment – typically 4 to 12 weeks (variable for every indication and country), represents an important yet often underutilized time to ensure patients are informed about all appropriate care options, including clinical trials. During this time, patients and their families are processing complex information, discussing treatment options, and making life-altering decisions.
Additionally, several structural factors limit the discussion of clinical trial options early in the patient journey. One key challenge lies in the tumor board process, where treatment plans are often discussed among multidisciplinary teams. While tumor boards play an essential role in determining the best treatment approach, they may also face limitations that affect trial awareness and referrals, such as:
- Potential conflicts of interest and ethical considerations when recommending experimental options
- Limited awareness or access to relevant trials, especially across different institutions
- Resource constraints that make it difficult to explore trial eligibility in depth
- Time pressure during case discussions
As a result, many patients begin standard treatment without ever learning about potentially relevant clinical trials
Clariness’ OncoCONNECT solution
To address these challenges, Clariness has developed OncoCONNECT, an innovative solution designed to support patients and healthcare professionals during this critical decision-making window.
OncoCONNECT leverages specifically trained oncology companions (oncologists) who act as trusted navigators throughout the journey to potential clinical trial enrollment. Their role is not to replace the physician but to support patients with clear information, guidance, and coordination, ensuring that clinical trials become a visible and understandable option.
By bridging the gap between diagnosis, treatment decisions, and trial opportunities, OncoCONNECT helps patients explore all available options while reducing the operational burden on physicians and research sites.
How Onco-Companions support the journey
Through OncoCONNECT, oncology companions provide personalized support at several key points in the patient journey.
Building trust: Receiving a cancer diagnosis is overwhelming, and many patients struggle to process the information they receive during early consultations. Oncology companions create a safe and supportive environment where patients can openly discuss their fears, hopes, and hesitations. This trust-building step allows patients to feel heard and supported while exploring their options.
Presenting clinical trial opportunities: Oncology companions help identify relevant studies and present available options in a clear and accessible way. By introducing clinical trials as one of several possible paths, patients gain a stronger sense of control and empowerment in their treatment decisions. Rather than feeling pressured, patients are encouraged to explore all available possibilities.
Simplifying complex medical information: Clinical research terminology and oncology treatment pathways can be difficult to understand. Oncology companions help translate complex medical language into clear, patient-friendly explanations, allowing patients to better understand their choices and ask informed questions.
Supporting family discussions: Treatment decisions often involve close family members. Oncology companions provide patients with educational and informational materials that help the conversation with their family and loved ones.
Connecting patients with HCPs: OncoCONNECT also helps coordinate communication between patients and their care teams. Oncology companions can organize joint discussions with healthcare professionals to explore potential trial opportunities and clarify medical questions.
Facilitating site connections: When a clinical trial appears to be a potential option, oncology companions help connect both patients and healthcare providers with appropriate research sites or investigators, ensuring a smooth transition from information gathering to potential enrollment.

Benefits for patients, physicians and sites
OncoCONNECT is designed to support the entirety of the ecosystem. For patients, it makes the clinical trial journey easier to understand and navigate during an emotionally challenging time. For healthcare professionals, it provides additional support in discussing complex treatment options without adding pressure to already limited consultation time. For trial sites, it helps ensure that patients who arrive for screening are better informed and more prepared, helping to reduce site burden and improve the efficiency of recruitment processes.
Closing note
Oncology (and rare diseases) recruitment is complex, but many of the challenges can be addressed by engaging patients earlier and supporting them through the decision-making process. Clariness’ new OncoCONNECT solution is designed to bridge the gap between diagnosis, treatment planning and clinical trial opportunities. Thus, helping patients, physicians, and research sites navigate this critical moment more effectively.
Take a look at our experience in patient recruitment for oncology clinical trials: Breast Cancer and Leukemia.
With more than 20 years of experience supporting oncology and rare disease trials, Clariness has partnered with sponsors and research sites worldwide to improve recruitment outcomes. To date, we have supported over 8,000 clinical trial sites and helped randomize more than 28,000 patients across complex studies.
Ready to accelerate recruitment in your oncology trial?
Speak with our experts to see how Clariness and OncoCONNECT can help you find, engage, and enroll the right patients faster.
Author: Mercy Hapsiba, Content Marketing Specialist | Post Date: 13.03.2026