COPD patient recruitment for clinical trials
Need help accelerating your study?
The number of people diagnosed with Chronic Obstructive Pulmonary Disease has grown by 27% over the last decade. With 15 million people diagnosed in the USA, and 115,000 new diagnosis yearly in the United Kingdom alone, the progressive lung disease leads to 3.3 million deaths annually worldwide.
At the same time, recruitment for COPD studies remains a struggle. According to Battaglia, only 1 in 4 patients are currently able to participate in clinical trials. Having successfully supported 30+ COPD trials around the world, we know how to overcome barriers to patient enrollment. To learn how we’ve supported sponsors with their studies, discover our recent case studies.

Reach COPD patients across the globe
As inclusion and exclusion criteria for COPD studies are often very strict, it is important to reach patients who are more likely to qualify and remain in the study.
Two of our large-scale patient surveys, with 4,500+ and 10,500+ participants respectively, showed us that 56% of our respondents would likely consider participating in a COPD clinical trial. In the larger survey, we engaged with a substantial number of at-risk and undiagnosed smokers with COPD-related symptoms who told us that it would be worth reaching out to them regarding studies aimed at those with mild-to-moderate COPD.
Our experience with patient recruitment for COPD studies has proven that a combination of digital patient outreach, lay-friendly study explanations, and study specific pre-screeners are able to overcome many of the problems faced when recruiting COPD patients for clinical trials.
Special screening techniques
Our data-driven digital outreach campaigns for COPD patient recruitment is focused on generating high volumes of traffic to our customized screeners. We know that for COPD, screening failures can occur even with a highly customized pre-screener.
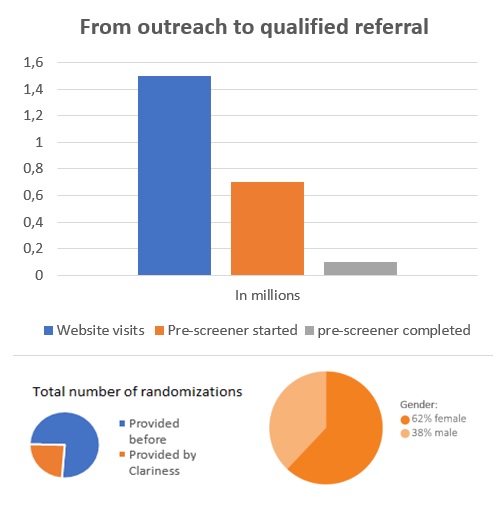
Seen here is an example of one campaign for 130+ research centers throughout Germany that drove 1.5 million visits to our pre-screener page. A total of 10,000 people completed the pre-screener successfully. The pre-screener consisted of multiple questions assessing medical eligibility, covering diagnosis and symptoms, that were based on the COPD definition of symptoms by the Global Initiative for COPD (GOLD).

Find patients aligned to your protocol
Our Medical Director reviews your protocol and aligns it with our patient recruitment team’s objectives and tactics. This ensures that patients referred to your research center fit the requirements of your study.
Our medical team then creates the dedicated study and indication pages and pre-screeners. They then develop a script for a secondary telephone screener. This ensures that any patients who successfully completed the online pre-screener, further qualify for your study’s protocol requirements.
We provide extra patient-friendly content that explains clinical trials, the regulations, safety measures, and your trial’s process. This helps ensure that patients who ultimately are randomized, are less likely to drop-out during the trial.
.
We understand that no two COPD studies are the same
From social advert creatives, to screeners and site services, we customize and align all materials to your target audience.
We understand the impacts of local culture, languages, and participation willingness. With this knowledge, we adapt to ensure we refer patients that meet your protocol’s requirements.
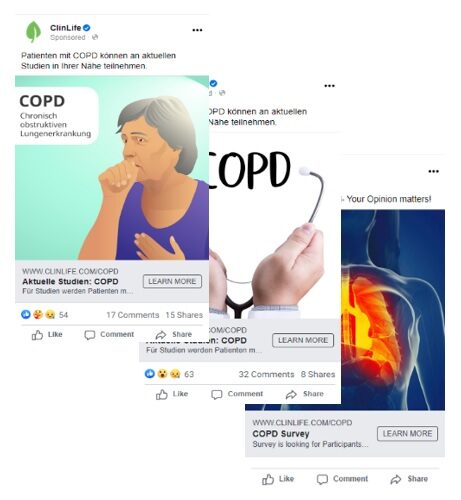
Seen here are three different creatives used within the same market, but with different COPD targets at the focus of each campaign.

Addressing patient diversity
Historically, marginalized populations are underrepresented in COPD clinical trials – despite evidence that these populations may be at increased risk. These include patients that identify as racial or ethnic minorities, or those with low socioeconomic status. As <50% of smokers will develop COPD during their lifetime, several environmental and genetic factors interact to determine whether a person develops COPD. Therefore, for a new treatment to be successful, having a diverse patient population is particularly important in COPD trials.
Informed by Patient Insights, our COPD patient recruitment and retention services allow us to reach a more diverse population. We use digital tools and multiple communication channels to reduce physical barriers to site access, create patient-friendly study materials to increase health literacy, and tailor strategies to local communities and cultures.
By understanding patient needs, we can overcome the unique barriers these patients face when joining clinical trials.

Leveraging decentralized solutions in your COPD studies
Embracing new decentralized clinical trial (DCT) solutions in your clinical trial improves the diagnosis and assessment of chronic conditions, such as COPD.
Recent research shows that wearables, unobtrusive sensors, or other digital technologies enables continuous real-world data collection that captures subtle changes in the health of patients. These technologies also reduce patient burden by limiting their travel and time commitments during a study. Digital technologies also enhance patient care and safety, while providing caregivers with an additional tool to look after their patients.
We can help you adopt new approaches and transform your trial by, for example, navigating regulatory changes, creating study materials for patients and caregivers to better understand new technologies, and providing intensified support to sites to reduce site burden.
Our experience with hybrid and DCTs can be applied directly to your study.
Our digital targeting techniques
We have four key ways of targeting for COPD patient recruitment.
Location and capacity targeting
Users living within a predesignated radius around active sites are targeted with ads that are activated and deactivated based on site referral volume.
Interest-based targeting
Users searching or engaging (e.g., likes, shares, comments, etc.) with COPD-indication related content and COPD stage-based symptoms.
Demographic targeting and pre-screener
Gender, age, and other demographics indicators are used as filters to target patients that likely fit the study criteria. An patient-centric pre-screener can furthermore help patients find out of they qualify for the study, for example by the number of migraine days in one month.
Behavioral targeting
By continuously analyzing the (anonymized) behavior of those successfully enrolled, we are able to better understand patients. Using this information, we reach potential patients that show similar behaviors.