patient recruitment
Bridging the gap in atopic dermatitis: addressing unmet needs
Currently, there’s no cure for atopic dermatitis. While recent biologic treatments help ease symptoms and reduce flare-ups, patients still face high unmet needs and significant impacts on quality of life.
Having supported 20+ atopic dermatitis trials (including both adult and adolescent patients), we know how to find the right candidates for your study.
finding the right patients
We can reach atopic dermatitis patients of all ages
As the most common skin disease in children, atopic dermatitis has historically been considered a pediatric disease, with a prevalence of up to 20% in certain countries. However, the condition can develop at any age: 1 in 4 adults report adult-onset of initial symptoms, with an estimated global prevalence of 1-10% in adults. Additionally, recent studies suggest a second peak in incidence after 60 years of age.
Despite being disproportionately affected, both children and older adults are underrepresented in atopic dermatitis clinical trials due to, for example, difficulties in approval from government regulatory bodies and strict I/E criteria. In the USA, the FDA only approves certain medications in different pediatric groups after initial approval in adults; while a systematic review of inclusion criteria in clinical trials on systemic atopic dermatitis treatments found that 34% of trials explicitly excluded older adults (Lam et al., 2020).
That’s why at Clariness, we target both patients and caregivers and have successfully recruited adolescents for atopic dermatitis clinical trials. In a recent atopic dermatitis patient insights survey (with 4,959 respondents), we were able to reach patients and caregivers of all ages.

A global success story for an atopic dermatitis study
Meet our Senior Project Manager, Renée Werner, who successfully led a global atopic dermatitis patient recruitment campaign involving seven protocols designed to recruit adults and adolescents from 18 countries across 260 sites.
Watch our video to see how we contributed 27% of all randomized patients to this study.
How we can help
Our digital targeting techniques
We have four ways of targeting patients and caregivers when performing atopic dermatitis patient recruitment, and continuously review performance to optimize engagement and trial registrations.
Demographic
- Gender, age, and other demographics indicators are used as filters to target patients that likely fit the study criteria, with these aspects included in each and every one of our screeners.
Location
- Patients living within a predesignated radius around active sites are targeted with a variety of advertisements in the local language and based on the local patient profile, that are activated and deactivated based on site referral volume.
Interest
- Patients searching or engaging (e.g., likes, shares, comments, etc.) with content related to atopic dermatitis, eczema, allergy asthma, dermatology, and skin problems are targeted with social media outreach.
Database
- We continuously analyze the behavior of those who did, and didn’t register, for trials to target patients with similar behaviors to boost conversions and improve ROI.
unique solutions
Our unique screening methods
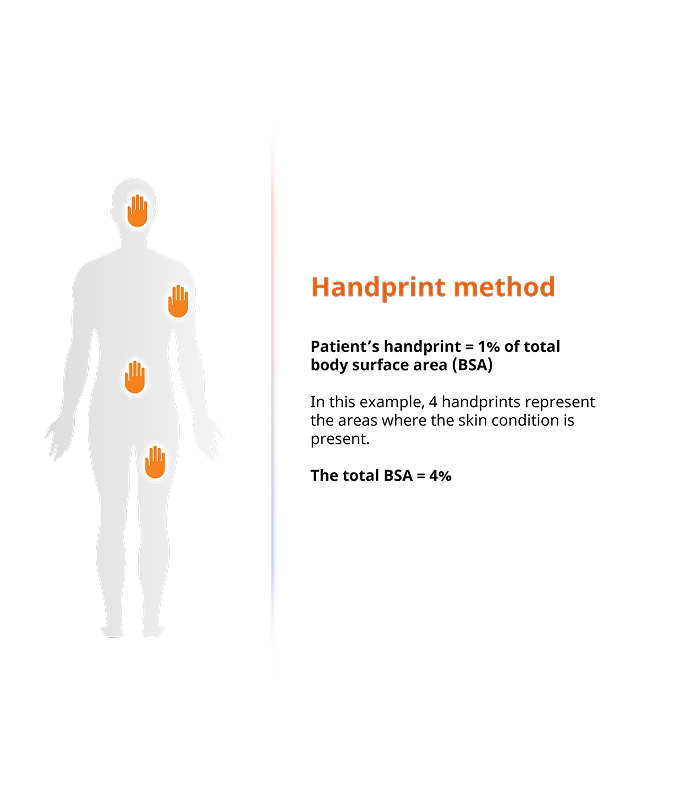
Accurately assessing body surface area (BSA) affected by atopic dermatitis has long been a challenge for patients.
To address this, Clariness developed a unique handprint screening technique, enabling patients to quickly and accurately self-identify symptom severity during phone and online screeners. This customizable approach, tailored to trial protocols, streamlines patient referrals to sites and boosts enrollment rates.
Want to learn more about our experience in atopic dermatitis?
tailored approach
Finding patients aligned to your protocol
Patients with atopic dermatitis experience unpredictable and highly variable signs and symptoms. Some studies suggest that >40% of patients with atopic dermatitis suffer from moderate to severe disease. Those with moderate-to-severe atopic dermatitis are also more likely to have higher medical costs, miss work, and be less satisfied with their treatment.
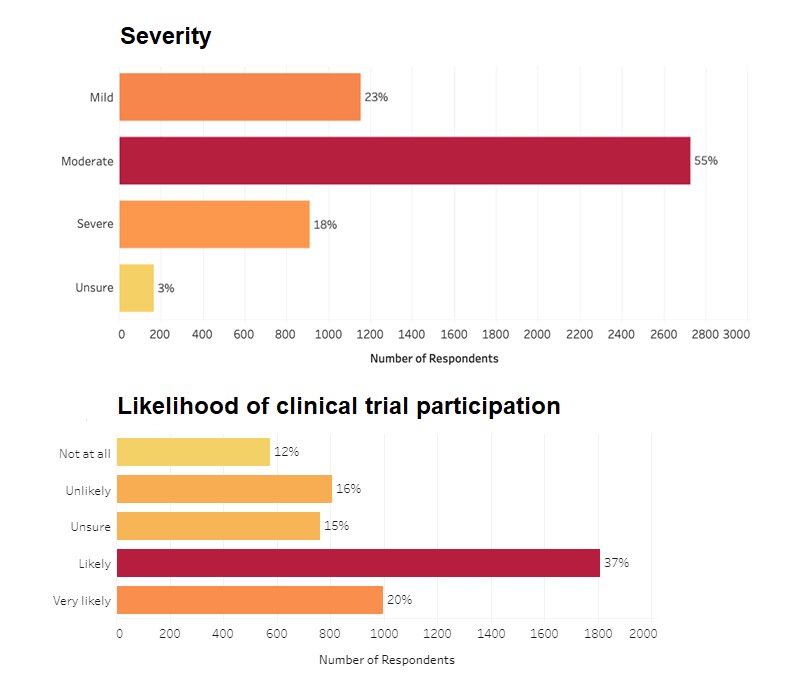
Our recent atopic dermatitis patient survey found that 55% of respondents (out of 4,959) rated their atopic dermatitis as moderate, with 18% rating it as severe. Additionally, >50% of respondents would consider participating in an atopic dermatitis clinical trial, with 20% indicating they are “very likely” and 36% reporting they are “likely” to do so.
Our survey results show that we can reach patients with mild to severe stages of disease who are interested in clinical trial participation. Our Chief Medical Officer will review your protocol and align with our patient recruitment team to develop methods that will ensure successful patient recruitment.
targeted outreach
We understand that no two atopic dermatitis studies are the same
From social advertising to screeners and site services, we customize and align all study services and materials to your target audience.
We understand the impacts of local culture, languages, and participation willingness and adapt accordingly to ensure we refer patients that meet your protocol’s requirements.
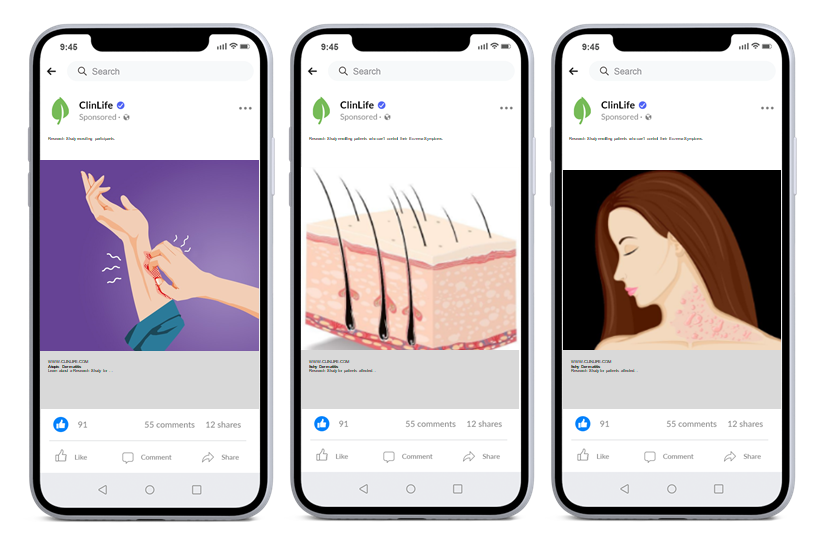
Here are three different advertising creatives used within the same market, but with different targets at the focus of each campaign. This allows us to capture the attention of a broader range of patients and caregivers for your clinical trial.
our experience
Download our atopic dermatitis case studies
- Total investment per patient 50% lower than planned
- Recruitment targets exceeded using 73% of budget
- Exceeded patient targets using 50% of budget
- Our campaign generated >365,000 ClinLife website visits
Please note that we will not share your data with any 3rd parties, data submitted will be used in accordance with our data privacy policy.