Since late 2021, there has been an increasing amount of calls from clinical industry leaders, pharmaceutical journalists, and even regulatory leaders such as the FDA, to act in order to improve participation rates in clinical trials. After the COVID-19 crisis and vaccine trials initially led to optimism about public awareness and willingness to participate, this turned out to be only a short-term boost as increased competition and patient demands led to increased and costly trial delays.
Indeed, reaching a high number of patients likely to match the study criteria and randomizing them remain the main challenge for clinical trial organizers. For this reason, in this blog we will look at how our patient-centric clinical trial platform, ClinLife®, manages to reach up to 6 times more patients in Germany than the platforms of leading pharmaceutical companies, and public databases.
Want more insights to support your studies?
How does ClinLife® reach more patients?
In 2022 over 89% of all Germans used the internet daily, on average for 3 hours in total. Studies have shown that these numbers are even higher for people with health problems. Accordingly, online medical searches are increasing both before and after doctor visits. In fact, Google (Dr. Google, as some researchers have called it) has become the primary way patients search for answers to their medical questions and has undeniably assumed a major role alongside medical professionals.
ClinLife® focusses on data-driven outreach over 40 channels to reach patients online. Based on their search interest, groups and other indication-specific categories this allows for a focus on patients likely to qualify for specific studies. Moreover, ClinLife® works together with patient organizations, health apps, and provides SEO-driven lay-friendly information in patient blogs and indication pages, also reaching patients searching for information through search engines.
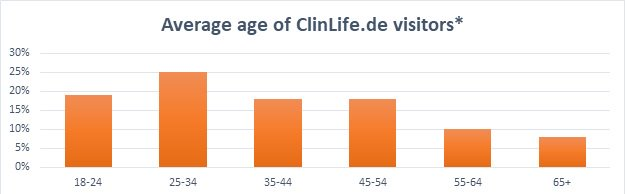
For this reason, ClinLife® is able to reach a more representative patient population compared to e.g. offline outreach or doctor referrals.
Table 2: Average age breakdown of ClinLife.de visitors
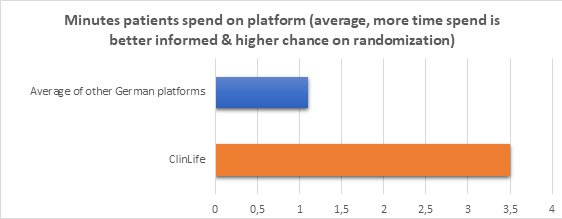
Why does ClinLife.de engage visitors significantly more than other platforms?
Compared to other portals, ClinLife® has a significantly higher engagement rate, with an average of 3.5 minutes per visitor, compared to below 1 minute for Germany’s largest sponsor portals. More informed patients are more likely to apply for a study, get successfully randomized, and stay in the study vs. visitors who quickly leave the website because they can’t find any relevant information.
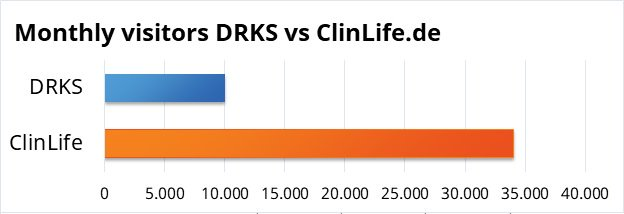
How does ClinLife® compare to the public German database DRKS (Deutsches Register Klinischer Studien)?
On ClinLife®, sponsors, CRO’s and university hospitals across Germany and the DACH-region list their studies on the same neutral environment. Patients select a study based on their preferences and if it matches their needs regardless of the specific sponsor. While this is similar to the public DRKS platform, ClinLife® has a user friendly interface that is easily navigable and accessible with options as text reading support, summarized study details and a patient-friendly pre-screener that helps patients find studies close to them.
How does Clariness’ ClinLife® patient portal compare to other third-party providers in patient recruitment?
Generally, patients can be reached over a variety of offline and digital channels but ultimately always come to the website before being enrolled to the study, either directly or through an innovative screener. Therefore, while the website visitor numbers don’t say everything, they give a good indication.
A crucial factor is however the difference in visitor amounts for different indications. Where healthy studies generally include a high number of participants and are easy to recruit for, rare diseases, oncology-studies or studies with very strict inclusion/exclusion criteria are more harder to reach a large patient population.
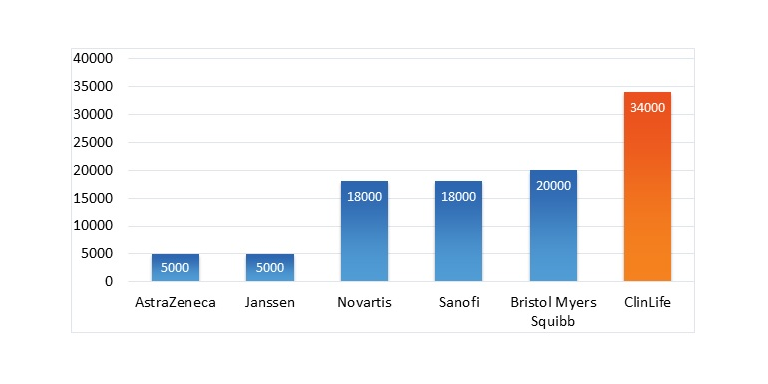
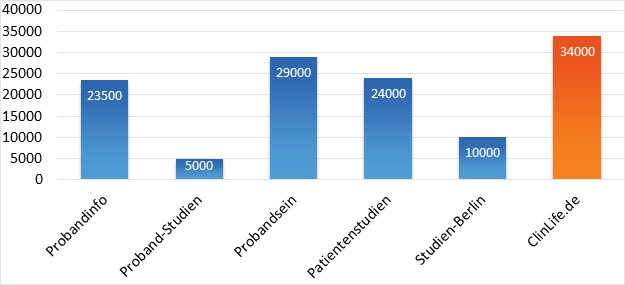
With this in mind, it is revealing to see how the visitor numbers of our German ClinLife® portal, which lists studies in over 40 indications, compare with those of other German third-party providers that focus mainly on health studies. The visitor numbers come from our internal tracking data and openly available sources such as SimilarWeb.
Comparison of the largest German portals for clinical trials. While most German portals focus exclusively on studies with healthy volunteers (Probanden in German), who can be recruited relatively easily in large numbers, ClinLife® lists studies from over 30 indications, including studies on rare diseases and oncology. (ClinLife® comparison numbers are based on numbers from both clinlife.de and clinlife.com/de/.
How could ClinLife® support your clinical trials and enhance recruitment rates?
Traditionally third-party providers are perceived as very costly methods of recruitment, but as mentioned, compared to clinical trial delays which can cost sponsors between $600K and $8M per day, the cost associated is often a fraction of these costs, typically a fraction of one day’s lost sales.
Clariness for example works on the basis of a flexible and protocol-based pricing model, giving options as covering risk shared, performance based and subscription-based model. The subscription model of the ClinLife® for example is a predictable and constant cost in the budget, providing a steady stream of referrals based on indication-based marketing. The advantage is that this does not require additional EC approval and can start almost immediately, as no study specific information is shown in the outreach and rather, patients are forwarded to a patient-friendly indication page and screener that helps the find suitable studies for their condition and in their proximity.
How does ClinLife® Registry compare to Clariness’ “active recruitment” project-based recruitment?
ClinLife® Registry can be complemented by Clariness’ project-based recruitment, that utilizes all the capabilities of Clariness from data-driven outreach methods based on study-specific criteria. This is suited for studies with very specific inclusion and exclusion criteria, or those towards the end a recruitment phase.
Why ClinLife® can accelerate your patient recruitment:
ClinLife® is a patient-centric clinical trials platform working on the basis of a marketplace principle, with sites, sponsors and CRO’s listing their studies on a easily navigable and neutral environment and patients finding matching studies in their proximity. Being developed in direct collaboration with patients, the platform helps patients to learn more about clinical trials and study procedures through lay-friendly information and collaborations as well as find matching studies through an innovative indication-based pre-screener.
We have successfully a 98% approval rating on first submission from EC/IRBs around the globe. Having successfully supported over 1,500+ studies and worked with 8,000+ sites in 54 countries, including >750 sites in Germany alone. We can support you from setting up the patient-friendly materials and getting EC approval, to creating study landing pages on our ClinLife® Portal. Enrollment can start within 2 weeks of contract signing.