Diabetes patient recruitment for clinical trials
Need help accelerating your study?
The number of patients diagnosed with diabetes rose from 108 million in 1980, to 463 million in 2021 worldwide. As a result, there has been a rapid increase in the number of clinical trials for both Type I and Type II diabetes. Yet, 50% of all diabetes studies are delayed because they fail to enroll sufficient patients. In fact, 4 out of 5 Type II diabetes trials do not reach their enrollment targets – significantly increasing waiting times to introduce new therapies, medicines, and interventions.
Having performed patient recruitment for 75+ local and global diabetes studies, leverage our knowledge and experience to improve the recruitment of your studies on Type I and Type II diabetes. To learn how we’ve supported sponsors with their studies, discover our recent case studies.
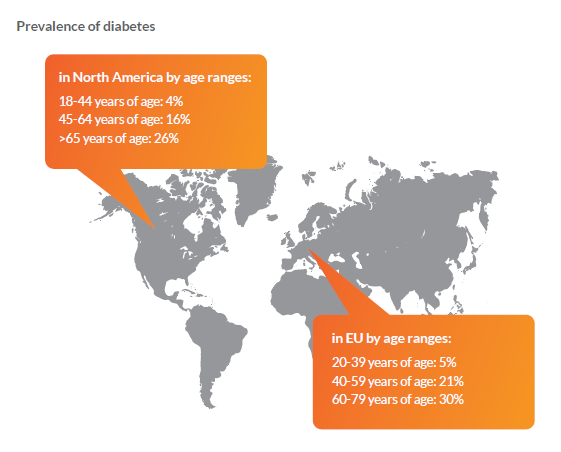
Challenges when recruiting patients for diabetes studies
- 50% (232 million) of people with diabetes are undiagnosed (according to the International Diabetes Federation). Therefore, it is hard to reach patients without specific outreach methods.
- Newly diagnosed people often do not feel sick or in need of treatment. Studies show that many patients (especially from minority groups) do not know about diabetes symptoms due to less access to healthcare, education, and lower socioeconomic status. Another barrier is what researchers describe as ‘Diabetes Denial,’ meaning many patients ignore mild symptoms.
- Many trials do not provide patient-centric information. Details on inclusion and exclusion criteria, the goals and procedure of the study, and other information are often not provided in a patient-centric manner. This leads to lower sign-up and participation rates, as well as higher drop-out during a clinical trial.
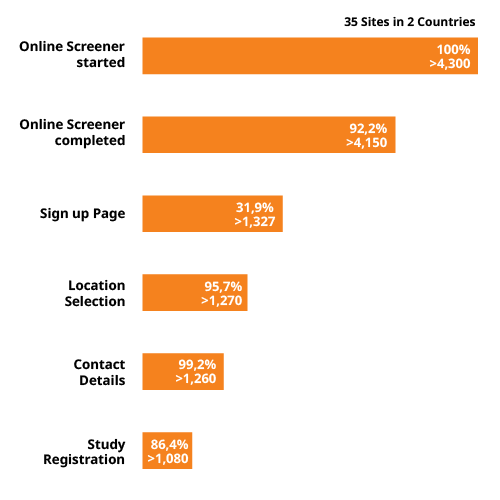
Special screening techniques
Our data-driven digital outreach campaign for diabetes studies focuses on generating high volumes of traffic to our customized screeners. Based on our experience, we know that screening failures will be high (especially for studies on Type II diabetes). Therefore, it is crucial to reach more people.
We developed a customized online screener that consists of multiple questions assessing medical eligibility, blood sugar criteria, and BMI criteria (BMI ≥ 24 & ≤ 40). For certain studies, we add questions that ensure willingness to participate, or the existence of hypoglycemia and malignancy within the last 5 years.
Our diabetes indication pages have received over 3 million unique visitors. Based on those who signed-up for trials listed on our patient portal, ClinLife, we learned that people who were newly diagnosed are more likely to participate in our studies. We then apply this information to our outreach campaigns.
Find patients aligned to your protocol
Our Chief Medical Officer reviews your protocol and aligns with our patient recruitment team’s objectives and tactics to ensure that patients referred to your research center fit the requirements of your study.
For diabetes trials, we know that removing or facilitating transportation barriers and updating the inclusion and exclusion criteria to better reflect patient populations can greatly improve registration and participation rates.
Our medical team then creates the dedicated study and indication pages and pre-screeners, as well as standardizes the script of the telephone screener. The telephone screener confirms that any patients who successfully completed the online pre-screener actually fulfills eligibility requirements of the study. This reduces the workload of and frustration felt by the study center and the patient.
We also provide patient-friendly content that explains clinical trials, regulations, safety measures, and specific processes during the clinical trial. This helps ensure that all patients, across different countries and age ranges, are better informed and less likely to drop-out during the trial.

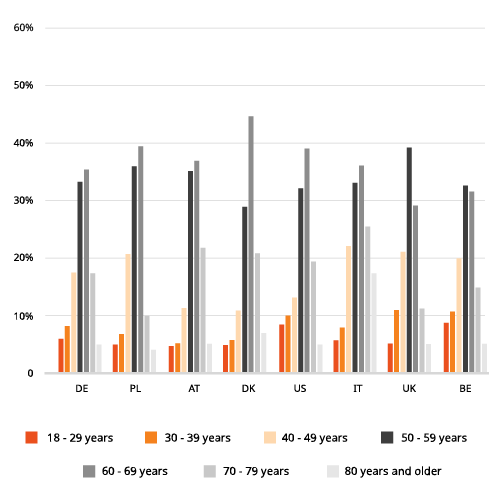
Addressing patient diversity
Historically, minority populations are underrepresented in clinical trials for diabetes, despite the fact that diabetes prevalence is higher in minority populations, as for example in Hispanic, Native American, and African American communities. Similarly in Europe, ethnic minorities are more often affected by diabetes, as well as people with a lower socioeconomic status.
Informed by Patient Insights, our diabetes patient recruitment and retention services allow us to reach a more diverse patient population. We use digital tools and multiple communication channels to reduce physical barriers to site access, create patient-friendly study materials to increase health literacy, and tailor strategies to local communities and cultures.
By understanding patient needs, we can overcome the unique barriers these patients face when joining clinical trials.

Leveraging decentralized solutions in your diabetes studies
Diabetes is one of the most common therapy areas for decentralized clinical trials (DCTs). Embracing new DCT solutions improves the participation rates of both Type I and II diabetes studies.
We can help you adopt new approaches and transform your trial by, for example, navigating regulatory changes, creating study materials for patients and caregivers to better understand new technologies, and providing intensified support to sites to reduce site burden.
Our experience with hybrid and DCTs can be applied directly to your study.
Our targeting techniques
We have four main ways of targeting for patient recruitment, addressing both patients with diabetes and caregivers.
Location & capacity targeting
We reach patients living in an area with a high prevalence of diabetes (and related causes and symptoms) and within a predesignated radius around active sites.
Interest-based targeting
Patients searching or engaging (e.g., likes, shares, comments, etc.) with diabetes-indication related content and symptoms are shown study advertisements.
Demographic targeting
Gender, age, ethnicity, social-economic class, location, and other demographics are used as filters to find patients (and their families) most likely to match the study.
Behavioral targeting
We email our large database of over 1.3 million registered users based on their location and indication. By continuously analyzing the (anonymized) behavior of those successfully enrolled, we can understand and reach similar people.

Download our diabetes case studies
Learn how we’ve supported sponsors with their patient recruitment for diabetes studies, across the globe, resulting in more engagement, more referrals, at less than the budgeted costs:
- Over 2,100 ClinLife referrals
- Successful targeting of patient subpopulation
- Our campaign generated >1.6 million ClinLife website visits
- RWE-driven strategy from 100k patients in 46 countries
Are you designing a diabetes clinical trial, or have one underway?
We can help you find the right patients, at the right time. To find out more about our heart failure patient recruitment, contact us.
